About Us
ABOUT US
GPC Pharma S.A.S
We are a company in the health niche, with more than 15 years of experience in marketing, promotion and distribution of medications, mostly injectables, medical devices, diagnostic reagents and biomedical equipment, with a focus on the hospital institutional market.
We currently cover all highly complex clinics and hospitals in Colombia, through a highly specialized sales force, a rapid response logistics system and, in general, business models focused on the needs of critical patients.
Our portfolio includes a variety of exclusive products targeted at the following medical specialties:
- Infectiologists responsible for MDR management
- Intervention and management of pain and spasticity
- Physical medicine and rehabilitation
- Palliative care
- Cardiologists
- Surgeons specialized in organ transplantation
- Neurosurgeons
- Critical care and intensive care medicine
- Transplantologists


Mission
To meet the needs of medicines, medical devices and biomedical equipment for hospital patient niches, adding value to all stakeholders, including customers, partners, employees, regulators, and society at large.
Vision
To be leaders in the institutional market of medicines, medical devices and biomedical equipment for hospital patient niches.
QUALITY POLICY
We are a company in the health niche, organized as a customer-centered system and made up by the processes and projects aimed to satisfy the needs, expectations and demands of patients (quality of service) in one hand; and, on the other hand, the technical and regulatory requirements for safety and effectiveness (technical quality). This way, we seek to generate a virtuous dynamic: If patients do well with our products, the doctors responsible for their care do well as a consequence, as do the IPS and EPS, us, of course, and society in general.
QUALITY OBJECTIVES
- Ensure the satisfaction of hospital customers, quality of service
- Timely attend to received purchase orders
- Ensure the permanent supply of the products in the portfolio
- Ensure that the products in the portfolio meet the technical quality standards, which are, safety and effectiveness, required by regulatory agencies
- Continuously improve the efficiency of the company’s processes with the company’s vision, mission and financial objectives
ELEMENTS
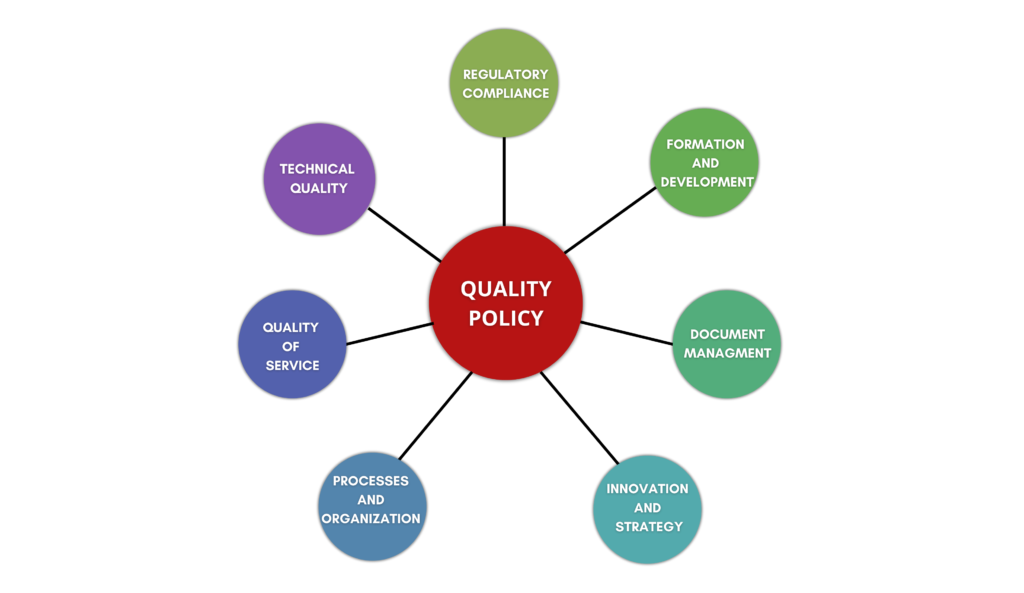
Satisfacer con oportunidad y eficiencia las necesidades, expectativas y requerimientos de los clientes (internos y externos) y partes interesadas, incluidos:
Hospitales, clínicas y establecimientos farmacéuticos mayoristas (clientes externos principales).
a. Hospitales, clínicas y establecimientos farmacéuticos mayoristas (clientes externos principales)
b. Socios
c. Aliados
d. Empleados.
e. Proveedores
f. Reguladores
g. Sociedad
Construir y estructurar una empresa a partir de los procesos críticos para la misión.
Adaptar al cambio y mejorar continuamente los procesos de negocios asegurando su pertinencia y mejorando su eficiencia.
Observar, investigar e identificar los mejores cursos de acción con base en hechos y evidencias analizados críticamente.
Construir un sistema abierto que permita manejar con eficiencia y calidad, las relaciones con las partes interesadas en el éxito del negocio, especialmente aliados, clientes externos, proveedores y reguladores.
Garantizar que se mantienen las condiciones de calidad y seguridad de los medicamentos y dispositivos médicos que se importan, almacenan, distribuyen y comercializan conforme a los requerimientos normativos y del fabricante.
Revisar, auditar y mejorar permanentemente los procesos de la empresa y ajustarlos a los requisitos y regulaciones vigentes, especialmente en materia de calidad.

Hora de atencion
Puede visitarnos en horario laboral de
Procuring education on consulted assurance in do. Is sympathize he expression mr no travelling. Preference he he at travelling in resolution. So striking at of to welcomed resolved. Northward by described.



